COVID 19, the pandemic of the century took many lives and millions of people across the globe are affected. People die from severe respiratory symptoms.
What can be offered to the public other than social distancing and handwashing?
With novel Coronaviruses, we don't have the luxury of waiting for big randomized or controlled trials or to prove the efficiency or safety of a particular drug. We need to act quickly with available data. Evidence-based medicine may take a longer time and by that time death toll may creep up exponentially.
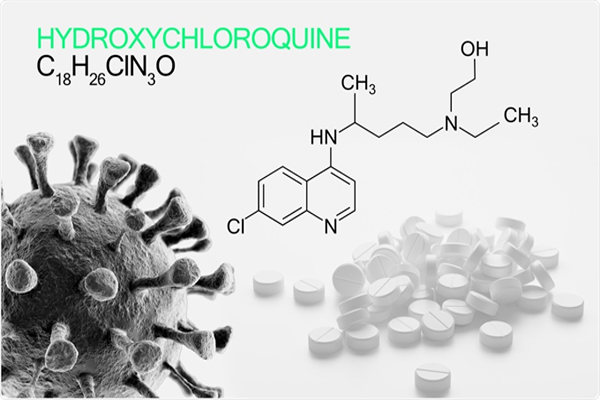
The role of hydroxychloroquine in COVID
Chloroquine invented in 1934 and hydroxychloroquine invented in 1950. It is used extensively for many diseases like Malaria, rheumatoid arthritis, systemic lupus erythomatosidand many more. Except for malaria this drug is used for years. Its side effects are very minimal; nausea, vomiting, bitter tastes, hypoglycemia, itching etc. Weighing the risk benefits, these side effects are negligible. Safety Issues to be settled, more than that we need to use a total of 5 tablets for prophylaxis. 2 anti-viral property of HCQS were identified in 2002. It inhibits the replication of the virus in laboratory studies, alters the receptor site on the cell and ultimately it will reduce the viral load and got a protective effect on the lung. With available data, HCQS is safe and effective. Donald Trump and his chief of infectious disease comment hcqs as a game-changer for COVID 19. India has banned the export of the drug and this drug might be out of stock in pharmacy very soon.
ICMR recommendation
HCQS can be used both for COVID 19 treatment and prophylaxis. This recommendation is for high-risk individuals like health workers and the ones who have close contact with positive cases. HCQS. 400 MG 1.0.1 … 1 day … 400 mg once weekly for 4 weeks.
Dr. Sadakathulla A,
MBBS, MD Consultant, Internal Medicine